TECNISTM Toric IOL
TECNISTM Toric IOL
To order this product, please contact your sales rep.
May not be available for all countries.
Instructional Guides
Instructional Guides
Instructional Guides
Excellent Outcomes for Patients with Astigmatism
Empower your patients’ vision with proven optical quality. The TECNISTM Toric IOL delivers sharp vision, good low-light performance and long-term sustainability for patients with astigmatism.1,2
Optimize your outcomes with the TECNIS Toric Calculator.
High-quality performance is just as critical for patients with astigmatism. The TECNISTM Toric IOL not only delivers excellent spherical correction but beautifully sharp vision as well.1
Excellent astigmatism correction meets superior image contrast:
- 94% of eyes achieved ≤ 0.50 diopters of residual refractive cylinder*2
- 88% of patients achieved monocular corrected distance visual acuity of 20/20 or better3
- Higher image contrast with TECNISTM Toric IOLs4
MTF at 50 c/mm White, 5 mm Pupil4
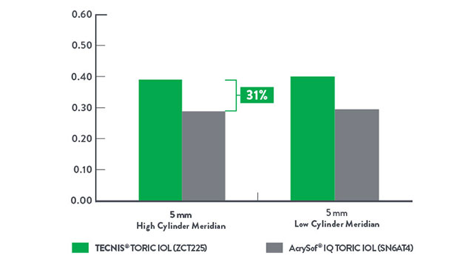
Up to 31% improved image contrast.
MTF at 50 c/mm White, 3 mm Pupil4
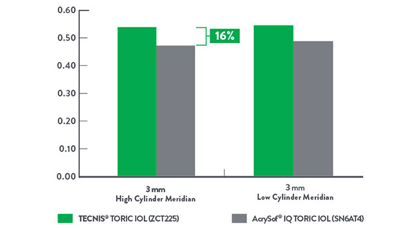
Up to 16% improved image contrast.
Modular Transfer Function (MTF) is a measure of the amount of contrast transferred by the optics in a visual system. The higher the MTF value, the more contrast transferred to the image, resulting in higher image contrast. AcrySof is a trademark of Novartis AG Corp.
TECNISTM Toric IOLs are designed for real-world conditions, empowering you to deliver high-confidence driving at night without distance glasses**3 — as well as good low-light performance for patients with astigmatism.
Difficulty With Driving at Night Without Glasses at 6 Months†3
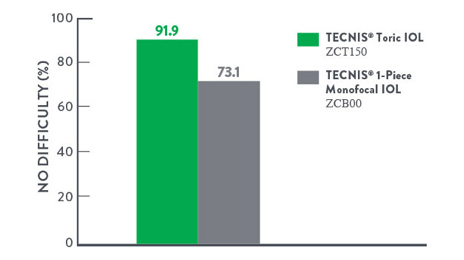
91.9% of patients (ZCT150) reported no difficulty with night driving — without glasses.
Trust your outcomes to an IOL that delivers exceptional postoperative rotational stability, stunning visual performance and high patient satisfaction.
2.74° mean axis change between baseline and 6 months.3
Unlike another leading IOL, TECNISTM IOL material is not associated
with glistenings,5-9 which can inhibit your patients’ vision. Glistenings
cause light scatter, which can result in a reduction in image contrast.6-10
97% of patients would elect to have the TECNISTM Toric IOL again.3
|
OPTIC CHARACTERISTICS1 |
||||||||||
|
Powers: |
+5.0 D to +34.0 D in 0.5 diopter increments. |
|||||||||
| Model Numbers: |
ZCT100
|
ZCT150 | ZCT225 | ZCT300 | ZCT375 | ZCT450 | ZCT525 | ZCT600 | ZCT700 | ZCT800 |
| Cylinder Powers – IOL Plane: | 1.00 D | 1.50 D | 2.25 D | 3.00 D | 3.75 D | 4.50 D | 5.25 D | 6.00 D | 7.00 D | 8.00 D |
| Cylinder Powers – Corneal Plane: | 0.69 D | 1.03 D | 1.54 D | 2.06 D | 2.57 D | 3.08 D | 3.60 D | 4.11 D | 4.80 D | 5.48 D |
|
Diameter: |
6.0 mm |
|||||||||
|
Shape: |
Biconvex, anterior toric aspheric surface, square optic edge |
|||||||||
|
Material: |
UV-absorbing hydrophobic acrylic |
|||||||||
|
Refractive Index: |
1.47 at 35° C |
|||||||||
|
Edge Design: |
ProTEC frosted, continuous 360° posterior square edge |
|||||||||
|
BIOMETRY |
CONTACT ULTRASOUND |
OPTICAL |
||||||||
|
A-Constant: |
118.8‡ |
119.3§ |
||||||||
|
Theoretical AC Depth: |
5.4 mm |
5.7 mm |
||||||||
|
Surgeon Factor:11 |
1.68 mm |
1.96 mm |
||||||||
|
HAPTIC CHARACTERISTICS1 |
||||||||||
|
Overall Diameter: |
13.0 mm |
|||||||||
|
Configuration: |
Tri-Fix design, modified C, integral with optic |
|||||||||
|
Material: |
UV-absorbing hydrophobic acrylic |
|||||||||
|
Design: |
Haptics offset from optic |
|||||||||
View full product specifications (PDF)
IOL INSERTION
Recommended insertion instruments:
- UNFOLDERTM Platinum 1 Series Delivery System: Intuitive push-and-twist, screw-style insertion designed to simplify lens implantation.
FOOTNOTES
*In a separate (IDE) study, 72.3% of ZCT150 eyes achieved ≤ 0.50 diopters of residual refractive cylinder. By comparison, 69.3% AcrySof® IQ Toric SA60T3 eyes achieved ≤ 0.50 D of residual refractive cylinder.
**As control eyes had ≤1.5 D of preoperative Kcyl, results for all toric eyes pooled are not to be compared to control values.
†Difficulty with certain activities without glasses at 6 months, bilateral subjects in the randomized control arm and open label arm safety population.
‡ Derived from clinical evaluation results of the TECNISTM 1-Piece IOL Platform for optical biometry.
§ A-Constant theoretically derived for ultrasound biometry.
REFERENCES
1. TECNISTM Toric 1-Piece IOL DfU OUS - Z311207, Rev. C, 11/2019. REF2020CT4322.
PP2022MLT4205

